Product Introduction:Victory Alloy High Durability Cobalt Based Wire
Victory Alloy cobalt based wire is a premium biomedical-grade cobalt-chromium alloy engineered specifically for the rigorous demands of orthopedic instruments and special medical components. Offering exceptional high wear resistance, high temperature stability, and outstanding corrosion protection, this high durability cobalt based wire outperforms traditional materials in surgical environments. Designed for manufacturers and medical device developers seeking reliable, long-lasting solutions in high-stress orthopedic applications, Victory Alloy wire ensures precision, biocompatibility, and extended service life under repeated sterilization, mechanical load, and bodily fluid exposure.


Features of Cobalt-Chromium Based Wire
Superior High Wear Resistance: Cobalt-chromium matrix with optimized carbide distribution provides outstanding abrasion resistance, ideal for cutting, drilling, and articulating surfaces in orthopedic instruments.
High Temperature Stability: Maintains high strength and structural integrity even at elevated temperatures during autoclave sterilization and high-speed surgical procedures.
Excellent High Corrosion Resistance: Chromium-rich passive layer delivers superior protection against bodily fluids, saline, and harsh disinfectants, far exceeding stainless steel in long-term biomedical use.
High Durability and Fatigue Strength: Non-magnetic cobalt based alloy offers exceptional tensile strength, stiffness, and fatigue resistance for repeated-use orthopedic tools and components.
Medical-Grade Biocompatibility: Fully compliant with ASTM F90 / ISO standards for implants and instruments; low nickel variants available for sensitive applications.
Precision Wire Forming: Available in custom diameters, shapes, and tempers for guide wires, cerclage cables, Kirschner wires, and specialized orthopedic instrument fabrication.
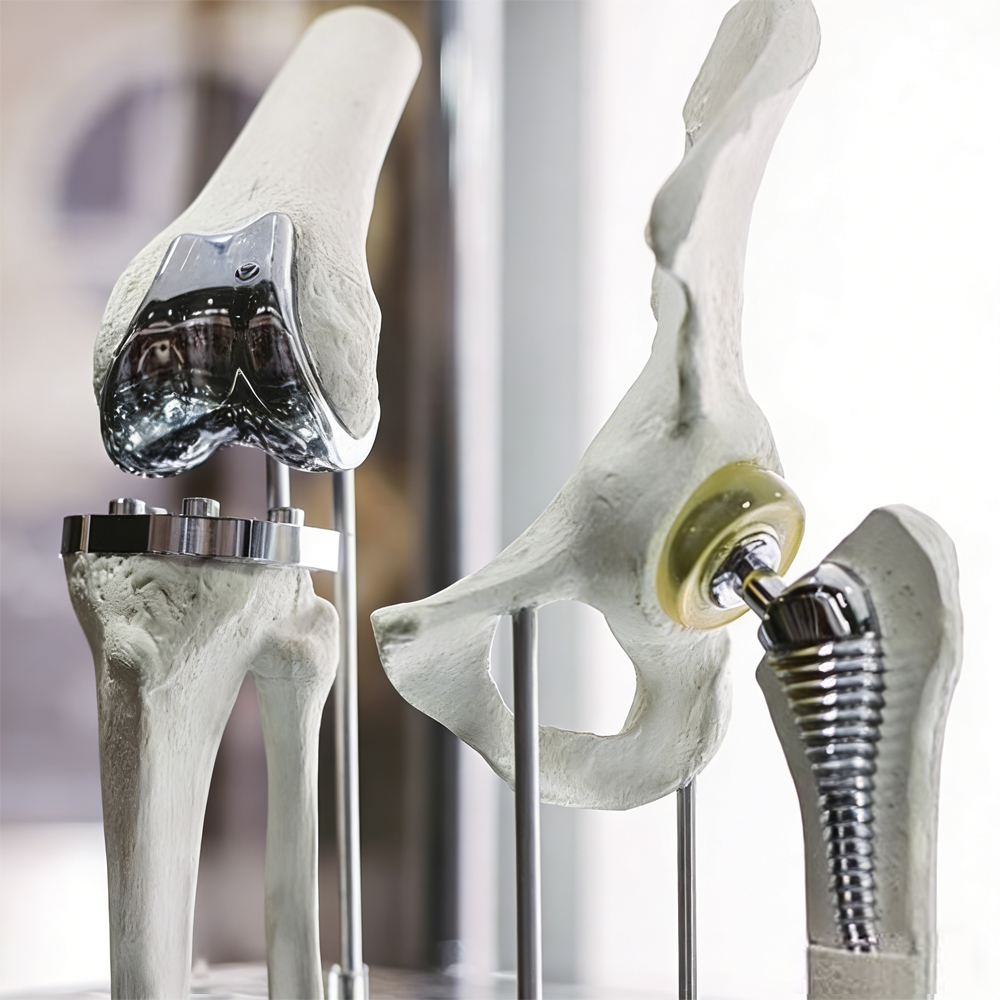
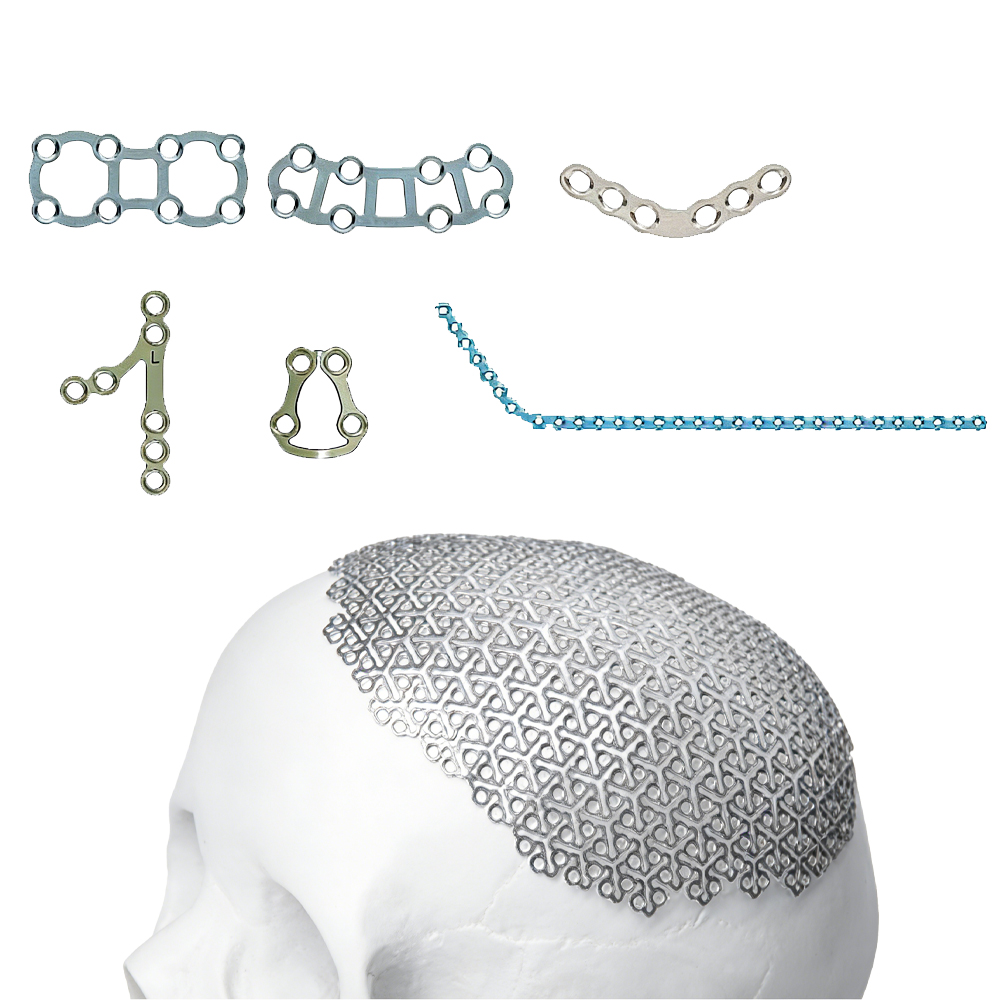
Applications in Special Medical Components
Medical Forceps Manufacturing: Jaws, tips, hinges, and spring components for reusable surgical forceps (hemostatic, tissue, biopsy, laparoscopic, and endoscopic forceps) requiring repeated sterilization and mechanical durability.
High-Corrosion Surgical Instruments: Blades, scissors, needle holders, and articulating mechanisms exposed to harsh cleaning protocols and bodily fluids.
Minimally Invasive & Endoscopic Devices: Guide wires, control cables, and micro-components in high-temperature, high-wear environments.
Dental & Orthopedic Tools: Precision wire for dental forceps, orthopedic retractors, and specialty instruments needing superior wear and corrosion resistance.
Custom Special Medical Components: High-performance parts for reusable devices in cardiology, urology, gynecology, and general surgery where standard stainless steel fails prematurely.

ABOUT US
Victory Alloy Co., Ltd. is a professional manufacturer specializing in medical alloy materials, with a dedicated focus on the research, development and production of high-performance biomedical alloys.
Our primary product range includes medical-grade titanium alloys, Nitinol (nickel-titanium alloy), cobalt-chromium (CoCr) alloys, platinum-iridium (Pt-Ir) alloys, and medical stainless steel. These high-quality alloys are widely applied in a broad spectrum of medical fields, such as cardiovascular stents, orthopedic implants, interventional guidewires, dental implants, surgical instruments, and disposable medical auxiliary devices. Boasting outstanding biocompatibility, strong corrosion resistance and excellent mechanical properties, our products have won consistent praise from customers across the board.

Client Visits
We welcome client visits to showcase our manufacturing and testing processes. Clients can explore our facility, discuss specific needs, and learn about our quality control methods. Our team ensures a personalized experience to meet your exact requirements for medical applications.

 Our Factory
Our Factory

Quality Testing of the Product
Our product undergoes strict testing for tensile strength, corrosion resistance, and biocompatibility to ensure reliable performance in joint implants. We use advanced methods to verify its safety and durability, and all products meet international medical standards for consistent, long-term use in the body.


FAQs on High Durability Cobalt Based Wire
Q1: What makes Victory Alloy cobalt based wire superior to stainless steel for orthopedic instruments?
A: Victory Alloy cobalt-chromium based wire offers significantly higher wear resistance, better high temperature stability during sterilization, and superior corrosion protection in bodily fluids compared to 316L stainless steel, while providing greater stiffness and fatigue life for long-term durability in special medical components.
Q2: Is this cobalt based wire suitable for high-temperature autoclave sterilization?
A: Yes. The alloy maintains excellent mechanical properties and structural integrity at elevated temperatures, making it ideal for repeated high-temperature sterilization cycles without degradation.
Q3: Does Victory Alloy cobalt based wire meet medical biocompatibility standards?
A: Absolutely. It complies with ASTM F90 and relevant ISO standards for biomedical use, ensuring full biocompatibility for orthopedic instruments and implantable-grade special medical components.
Q4: What diameters and custom options are available for orthopedic applications?
A: Available in a full range of precision diameters (0.1 mm to 5.0 mm+), round, flat, or shaped profiles, with custom tempers and surface finishes tailored for guide wires, cerclage cables, or instrument fabrication.
Q5: Why choose cobalt based wire for high-wear orthopedic instruments?
A: Its unique cobalt-chromium matrix delivers unmatched abrasion resistance and hardness, dramatically extending the service life of drills, reamers, and cutting instruments under continuous mechanical stress.
Q6: Is the wire corrosion-resistant in saline and bodily fluids?
A: Yes. The chromium oxide layer provides exceptional high corrosion resistance, preventing pitting and degradation even during prolonged exposure in surgical or in-vivo environments.










