Product Introduction:ASTM F2063 Superelastic Nitinol Springs
Our medical grade superelastic Nitinol springs (NiTi alloy) fully comply with ASTM F2063 standards, delivering exceptional superelasticity and biocompatibility for long-term implantable medical devices. Manufactured as an experienced OEM supplier, these superelastic Nitinol springs exhibit up to 8-10% recoverable strain at body temperature, enabling kink-resistant, self-expanding performance without permanent deformation.
Ideal for minimally invasive surgical applications, our implantable Nitinol components offer superior fatigue resistance (>10^7 cycles), corrosion resistance in physiological environments, and a stable titanium oxide layer for enhanced biocompatibility. As a professional China-based OEM Nitinol manufacturer, we provide custom superelastic Nitinol springs in various wire diameters, coil configurations, and transformation temperatures (Af tailored to body temp), ensuring reliable supply for global medical device companies.

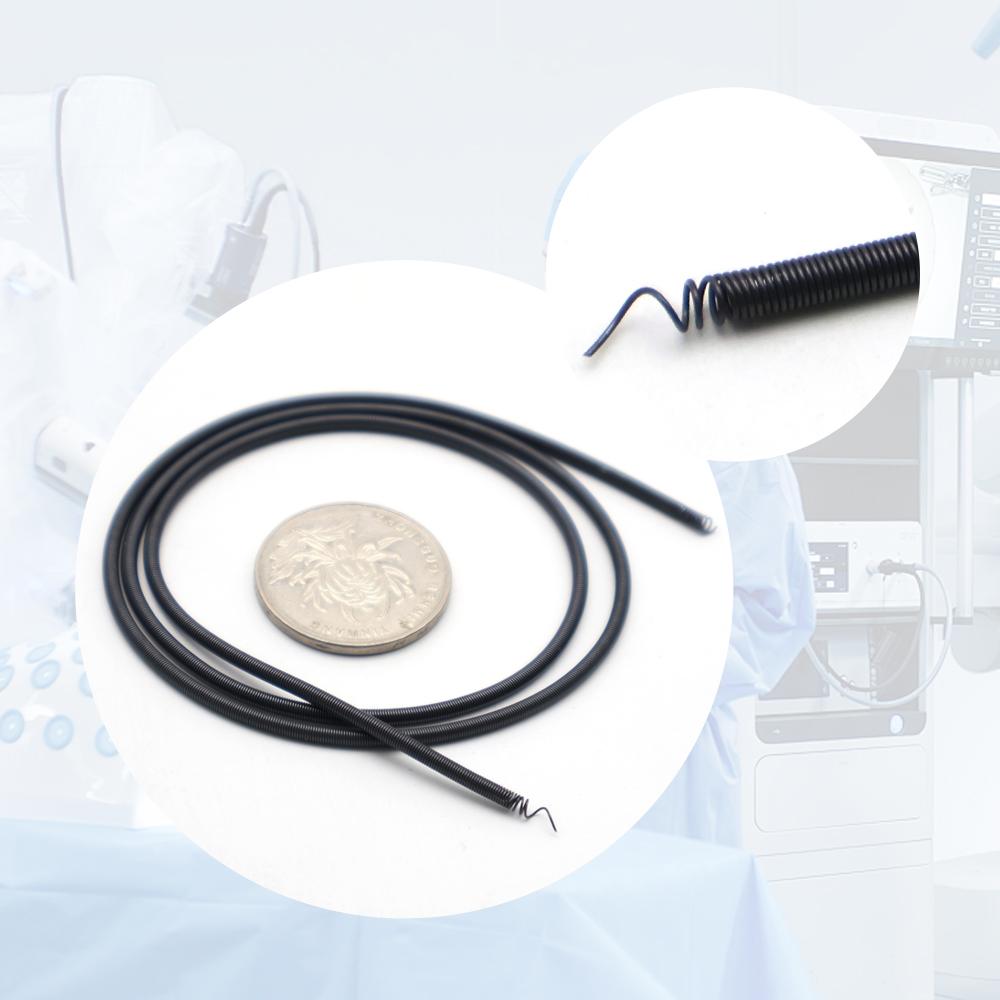
Features of ASTM F2063 Superelastic Nitinol Springs
Engineered for demanding implantable applications, our medical grade superelastic Nitinol springs feature:
Ultra-high superelasticity — recoverable strains up to 8-10% at body temperature (Af ~30-37°C) for kink-free deployment
Biocompatible & implantable safety — meets ASTM F2063 chemical composition (Ni 54.5-57 wt%), low inclusions, and passivation for long-term human implantation
Exceptional fatigue & corrosion resistance — withstands millions of cycles in corrosive bodily fluids, ideal for chronic implants
Custom OEM configurations — compression, extension, torsion, conical Nitinol springs; wire Ø 0.1–2.0 mm, custom pitch & length
Shape memory option available — combined superelastic/shape memory for advanced actuation in medical tools
Traceable & certified production — full material certification, cleanroom processing, and FDA/ISO-compliant quality system
Choose our superelastic Nitinol springs as your reliable implantable Nitinol components source from a dedicated OEM supplier.
Applications of ASTM F2063 Superelastic Nitinol Springs
Medical grade ASTM F2063 superelastic Nitinol springs are widely adopted in high-precision implantable and interventional devices worldwide:
Cardiovascular implants — self-expanding stent delivery systems, heart valve frames, atrial septal defect occluders
Orthopedic & spinal implants — bone anchors, spinal correction rods, dynamic stabilization springs
Minimally invasive surgery — retrieval baskets, snares, endoscopic clips, guidewire components
Urology & gastroenterology — ureteral stents, stone retrieval devices, esophageal stents
Neurovascular devices — embolization coils, aneurysm clips with superelastic recovery
Custom OEM medical tools — actuators, filters, and deployable structures requiring reliable expansion/compression
Partner with our implantable Nitinol OEM supplier for tailored superelastic Nitinol springs that meet stringent regulatory requirements in these critical fields.
ABOUT US
Victory Alloy Co., Ltd. is a professional manufacturer specializing in medical alloy materials, with a dedicated focus on the research, development and production of high-performance biomedical alloys.
Our primary product range includes medical-grade titanium alloys, Nitinol (nickel-titanium alloy), cobalt-chromium (CoCr) alloys, platinum-iridium (Pt-Ir) alloys, and medical stainless steel. These high-quality alloys are widely applied in a broad spectrum of medical fields, such as cardiovascular stents, orthopedic implants, interventional guidewires, dental implants, surgical instruments, and disposable medical auxiliary devices. Boasting outstanding biocompatibility, strong corrosion resistance and excellent mechanical properties, our products have won consistent praise from customers across the board.

Client Visits
We welcome client visits to showcase our manufacturing and testing processes. Clients can explore our facility, discuss specific needs, and learn about our quality control methods. Our team ensures a personalized experience to meet your exact requirements for medical applications.

 Our Factory
Our Factory

Quality Testing of the Product
Our product undergoes strict testing for tensile strength, corrosion resistance, and biocompatibility to ensure reliable performance in joint implants. We use advanced methods to verify its safety and durability, and all products meet international medical standards for consistent, long-term use in the body.


FAQs on ASTM F2063 Superelastic Nitinol Springs Supplier
Q1: What does ASTM F2063 compliance mean for superelastic Nitinol springs?
A: ASTM F2063 specifies chemical composition, mechanical properties, and processing for wrought NiTi alloys used in surgical implants—ensuring our medical grade superelastic Nitinol springs are safe, biocompatible, and reliable for long-term implantation.
Q2: Are your Nitinol springs suitable for permanent implantable medical devices?
A: Yes—our superelastic Nitinol springs feature high biocompatibility (titanium oxide surface), low nickel release, and proven fatigue life, making them ideal for chronic implants like stents, anchors, and occluders.
Q3: Can I order custom sizes and configurations of implantable Nitinol springs OEM?
A: Absolutely. As a specialized OEM supplier, we offer full customization: wire diameter, coil count, spring rate, Af temperature, compression/extension/torsion types, with rapid prototyping and scalable production.
Q4: What transformation temperature is recommended for body-temperature superelastic Nitinol springs?
A: For optimal superelastic performance in vivo, we recommend Af between 30–37°C (body temp range), ensuring full superelastic recovery without thermal activation.
Q5: Why choose your China OEM manufacturer for medical grade ASTM F2063 Nitinol springs?
A: We provide cost-effective, high-quality implantable Nitinol components with strict ASTM F2063 compliance, full traceability, cleanroom manufacturing, global certifications, and competitive OEM pricing—trusted by medical device innovators worldwide.









