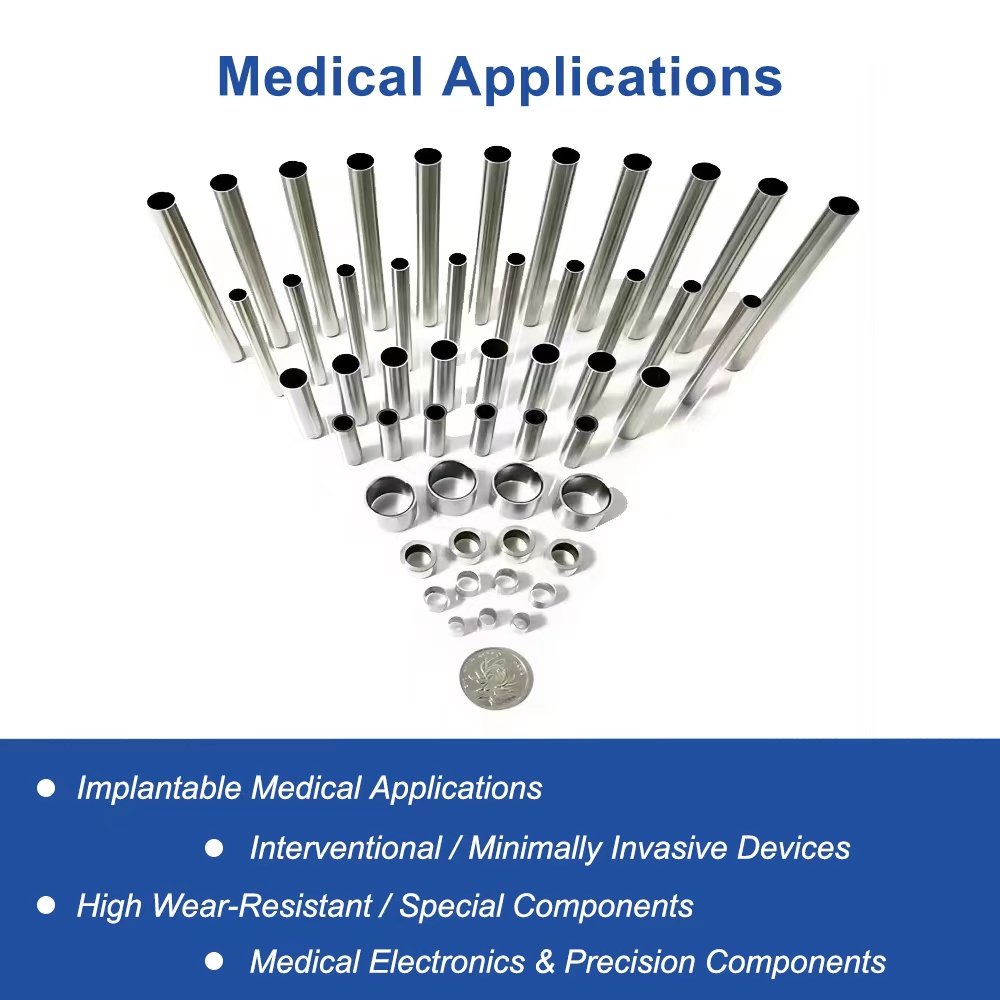
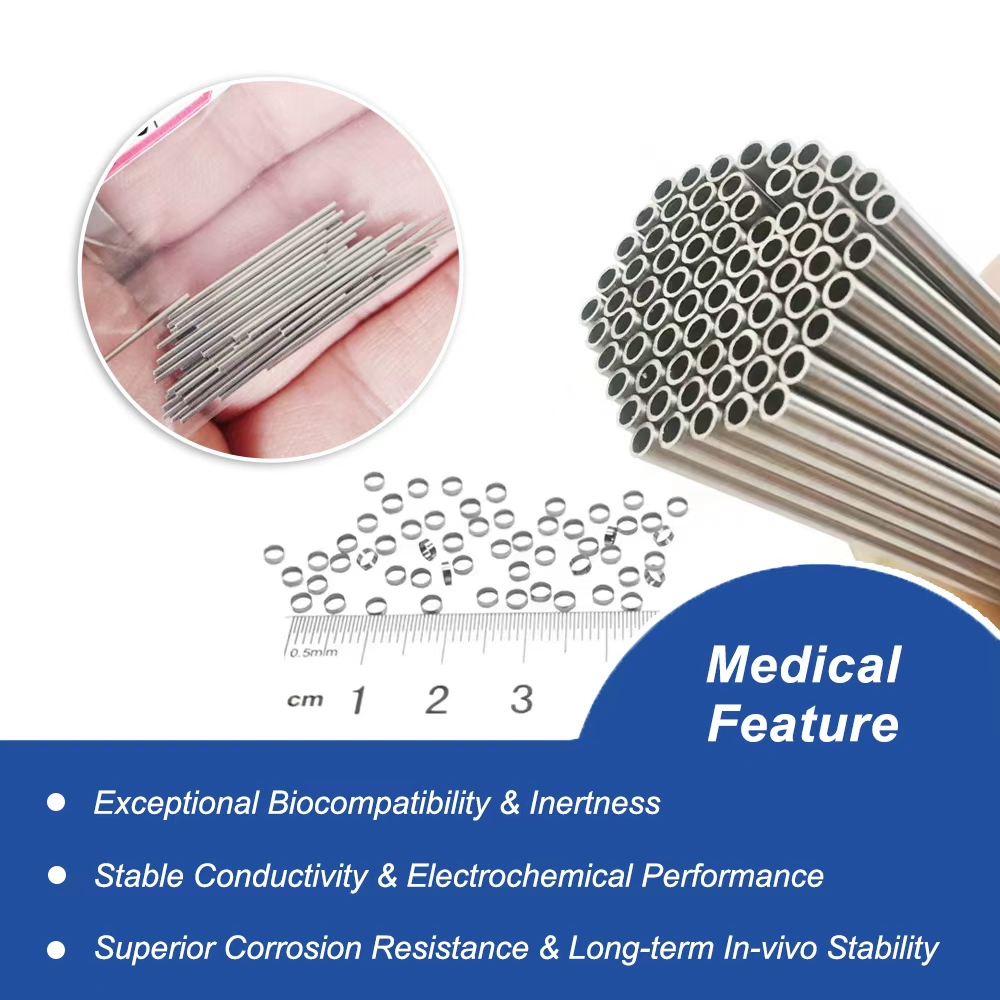
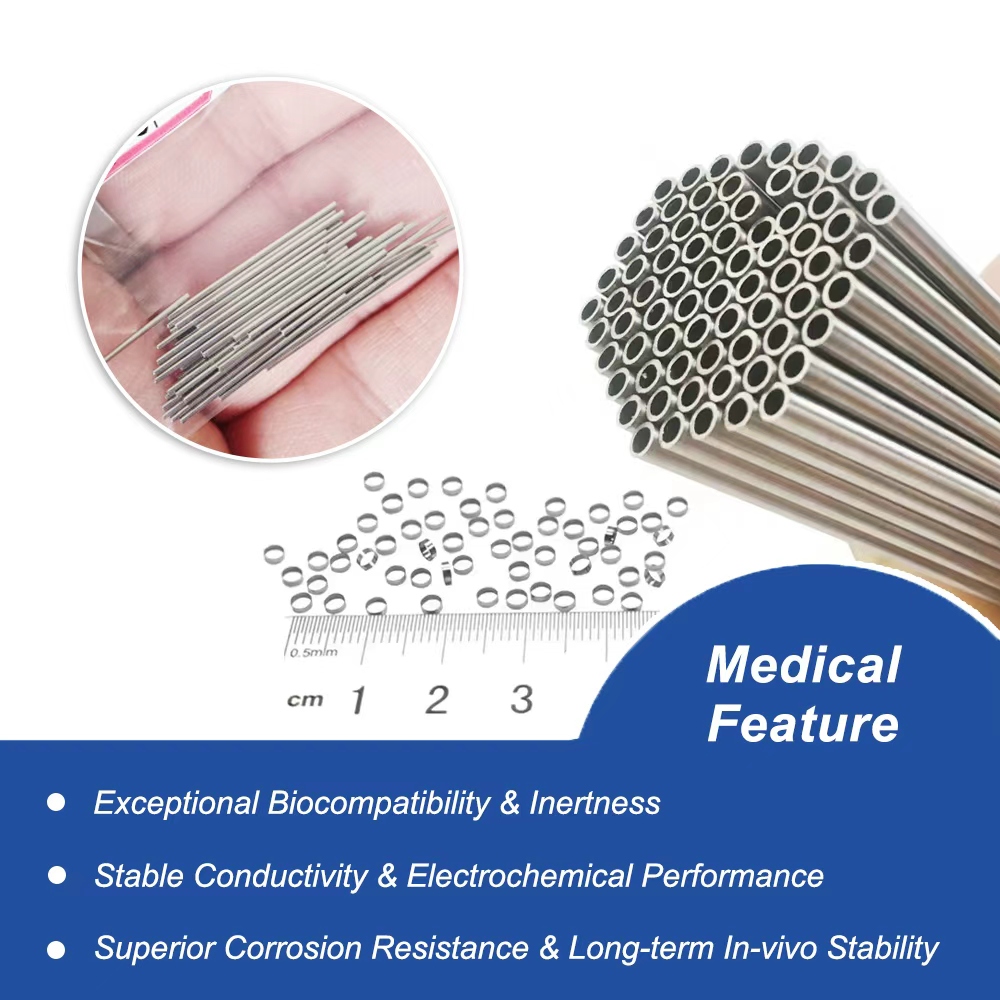
Overview of High-Strength Pt-Ir Alloy Tubes
Our premium biocompatible platinum iridium alloy tubes are engineered specifically for pacemaker components, providing essential materials for interventional cardiology and minimally invasive instruments. These Pt-Ir tubes combine the exceptional properties of platinum and iridium, ensuring biocompatibility, radiopacity, and mechanical strength critical for cardiac implants and implantable medical devices. Ideal for global markets including North America, Europe, and Asia-Pacific regions, these tubes support advanced pacemaker manufacturing, offering seamless integration into minimally invasive surgery tools and interventional devices. With a focus on medical tubing standards like ISO 10993 for biocompatibility testing, our alloy tubes minimize patient risks while maximizing device longevity in electrophysiology applications and heart rhythm management systems.

Features of Pt-Ir Alloy Tubes in Minimally Invasive Pacemaker Instruments
Superior Biocompatibility for Cardiac Implants: Compliant with biocompatibility standards, these platinum iridium tubes reduce immune responses, making them perfect for long-term implantable devices in interventional cardiology across global healthcare settings.
High Radiopacity for Interventional Imaging: Enhanced visibility under fluoroscopy and X-ray, aiding precise placement in minimally invasive procedures for pacemaker leads and components in electrophysiology labs worldwide.
Exceptional Corrosion Resistance in Medical Tubing: Pt-Ir alloy withstands bodily fluids and sterilization processes, ensuring durability for pacemaker manufacturing and minimally invasive instruments in diverse geographic markets like the US, EU, and emerging Asia-Pacific regions.
Precision Dimensions for Implantable Components: Customizable wall thickness and diameters (e.g., 0.1mm to 5mm) support intricate designs in cardiac rhythm devices, optimizing performance in interventional vascular applications.
Mechanical Strength for Heart Device Reliability: High tensile strength and fatigue resistance prevent breakage in dynamic environments, ideal for global suppliers of minimally invasive surgery tools and pacemaker accessories.
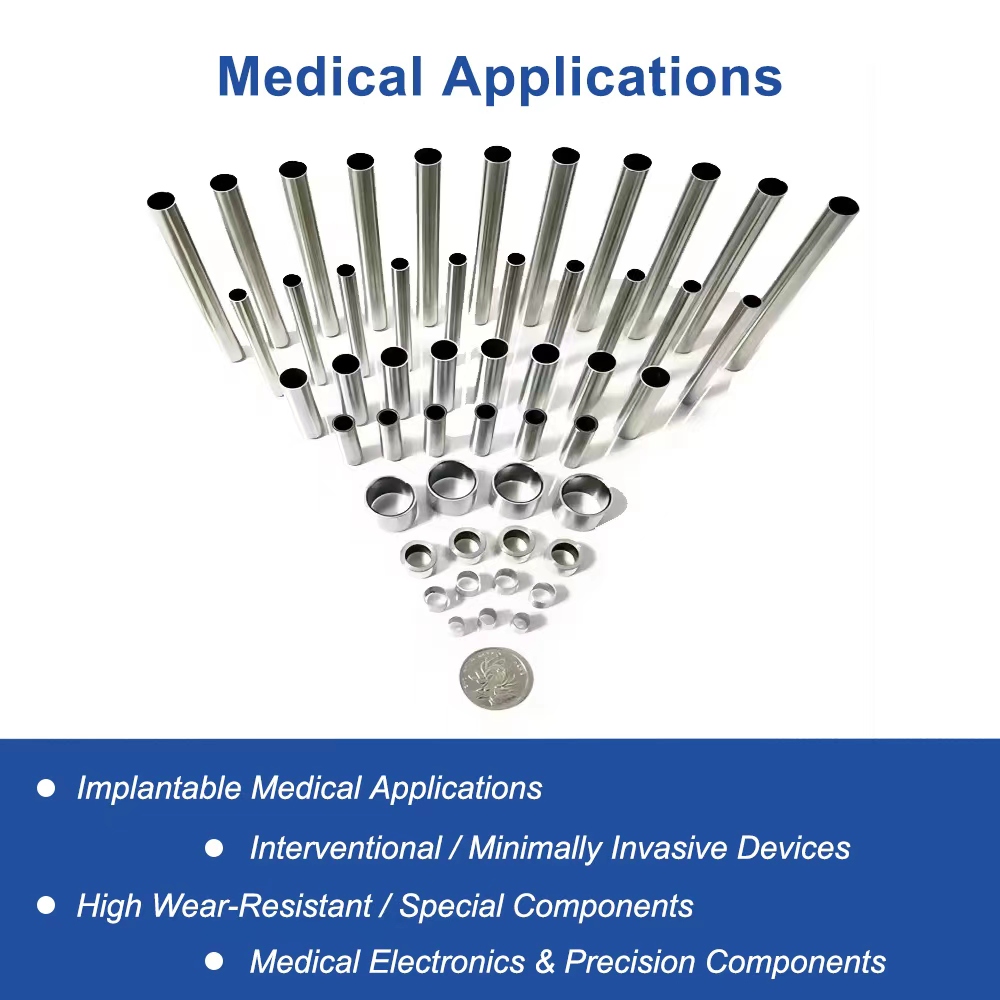
Applications of Pt-Ir Alloy Tubing in Minimally Invasive Medical Devices
Pacemaker Lead Manufacturing in Global Cardiology: Used as core components in pacemaker electrodes and leads, enhancing signal transmission for heart rhythm management in interventional procedures across North American and European hospitals.
Minimally Invasive Surgery Tools for Electrophysiology: Integral to catheters and guidewires in cardiac ablation and mapping, providing biocompatible support for precise interventions in Asia-Pacific medical centers.
Implantable Medical Devices for Vascular Access: Supports stent delivery systems and vascular grafts, ensuring safe, minimally invasive implantation in worldwide interventional radiology practices.
Cardiac Implant Components in Biocompatible Alloys: Essential for sensor housings and connectors in advanced pacemakers, promoting longevity in global markets focused on heart failure treatment and arrhythmia management.
Interventional Instruments for Pediatric Cardiology: Customized Pt-Ir tubes for smaller-scale devices, aiding minimally invasive treatments in international pediatric hospitals with emphasis on biocompatibility and safety.


ABOUT US
Victory Alloy Co., Ltd. is a professional manufacturer specializing in medical alloy materials, with a dedicated focus on the research, development and production of high-performance biomedical alloys.
Our primary product range includes medical-grade titanium alloys, Nitinol (nickel-titanium alloy), cobalt-chromium (CoCr) alloys, platinum-iridium (Pt-Ir) alloys, and medical stainless steel. These high-quality alloys are widely applied in a broad spectrum of medical fields, such as cardiovascular stents, orthopedic implants, interventional guidewires, dental implants, surgical instruments, and disposable medical auxiliary devices. Boasting outstanding biocompatibility, strong corrosion resistance and excellent mechanical properties, our products have won consistent praise from customers across the board.

Client Visits
We welcome client visits to showcase our manufacturing and testing processes. Clients can explore our facility, discuss specific needs, and learn about our quality control methods. Our team ensures a personalized experience to meet your exact requirements for medical applications.

 Our Factory
Our Factory

Quality Testing of the Product
Our product undergoes strict testing for tensile strength, corrosion resistance, and biocompatibility to ensure reliable performance in joint implants. We use advanced methods to verify its safety and durability, and all products meet international medical standards for consistent, long-term use in the body.


FAQs on Medical Grade Platinum-Iridium Tubes
1.What Makes Platinum Iridium Alloy Tubes Biocompatible for Pacemaker Components in Minimally Invasive Surgery?
Platinum iridium (Pt-Ir) alloy tubes are biocompatible due to their inert nature, passing ISO 10993 tests for cytotoxicity and sensitization. This ensures safe use in pacemaker components and interventional instruments, reducing risks in global cardiac procedures.
2.How Do Pt-Ir Alloy Tubes Enhance Radiopacity in Interventional Cardiology Devices Worldwide?
The high density of iridium in Pt-Ir tubes provides excellent radiopacity, allowing clear visualization during fluoroscopic-guided minimally invasive pacemaker implantations, beneficial for electrophysiology specialists in regions like the US and Europe.
3.Are These Biocompatible Tubes Customizable for Specific Pacemaker Manufacturing Needs in Asia-Pacific Markets?
Yes, our premium platinum iridium tubes offer customizable lengths, diameters, and finishes to meet diverse pacemaker component requirements in minimally invasive instruments, supporting global supply chains and regional medical device regulations.
4.What Corrosion Resistance Benefits Do Platinum Iridium Tubes Offer for Implantable Cardiac Instruments?
Pt-Ir alloy tubes exhibit superior resistance to corrosion from bodily fluids and electrolytes, extending the lifespan of pacemaker leads and interventional tools in harsh in-vivo environments across international healthcare systems.