Overview of Advanced Cobalt Chromium Tube
Our advanced cobalt chromium tube is precision-engineered as a high-performance drug-eluting stent platform in implantable medical applications. Crafted from medical grade CoCr alloy (conforming to ASTM F562 / L605 equivalents), this seamless tubing delivers unmatched mechanical properties for modern coronary stent manufacturing.
Compared to traditional stainless steel, this cobalt chromium alloy tube enables thinner struts without compromising structural integrity — reducing neointimal hyperplasia risk while enhancing deliverability, vessel conformability, and long-term patency in drug-eluting stent systems. Ideal for next-generation cardiovascular stents, it supports controlled drug release coatings and promotes superior endothelialization in percutaneous coronary interventions.

Features of Medical Grade Cobalt Chromium Tube
High Strength & Thin-Strut Capability — Superior yield strength (≈45% higher than 316L stainless steel) allows ultra-thin struts for improved flexibility and reduced restenosis in drug-eluting stent platforms.
Exceptional Biocompatibility — Proven low thrombogenicity and minimal inflammatory response; widely used in FDA/CE-approved implantable medical devices like coronary stents and valve frames.
Outstanding Fatigue Resistance — Engineered for >10^8 pulsatile cycles, ensuring durability under physiological loading in cardiovascular stent environments.
Enhanced Corrosion Resistance — Chemically inert CoCr alloy minimizes ion release (Co, Cr, Ni significantly reduced) and eliminates pitting in vascular applications.
Superior Radiopacity & MRI Compatibility — Higher density for excellent fluoroscopic visibility; non-ferromagnetic for safer MRI procedures.
Precision Seamless Manufacturing — Laser-cut friendly tubing with uniform wall thickness, smooth surface finish, and tight tolerances for consistent stent platform performance.

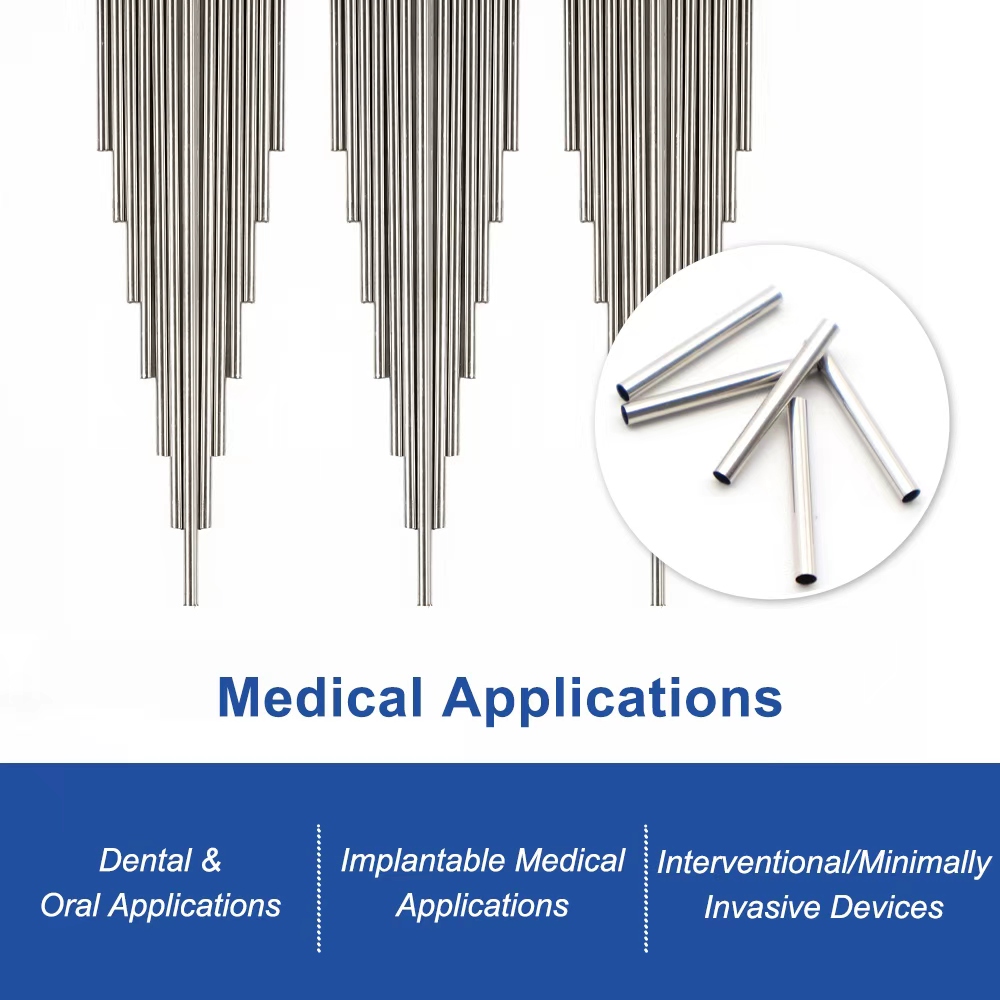
Applications of Implantable Medical Devices & Drug-Eluting Stent Platforms
This advanced cobalt chromium tube serves as the foundational platform material for:
Drug-eluting coronary stents (DES) — Enabling thinner struts, better apposition, and lower target lesion revascularization rates.
Bare-metal and bioresorbable polymer stent systems requiring high radial force and fatigue endurance.
Peripheral vascular stents and endovascular devices demanding corrosion resistance in dynamic anatomy.
Structural components in transcatheter heart valves, aneurysm clips, and other implantable cardiovascular devices.
Next-generation interventional cardiology solutions focused on long-term vessel patency and reduced in-stent restenosis.
ABOUT US
Victory Alloy Co., Ltd. is a professional manufacturer specializing in medical alloy materials, with a dedicated focus on the research, development and production of high-performance biomedical alloys.
Our primary product range includes medical-grade titanium alloys, Nitinol (nickel-titanium alloy), cobalt-chromium (CoCr) alloys, platinum-iridium (Pt-Ir) alloys, and medical stainless steel. These high-quality alloys are widely applied in a broad spectrum of medical fields, such as cardiovascular stents, orthopedic implants, interventional guidewires, dental implants, surgical instruments, and disposable medical auxiliary devices. Boasting outstanding biocompatibility, strong corrosion resistance and excellent mechanical properties, our products have won consistent praise from customers across the board.

Client Visits
We welcome client visits to showcase our manufacturing and testing processes. Clients can explore our facility, discuss specific needs, and learn about our quality control methods. Our team ensures a personalized experience to meet your exact requirements for medical applications.

 Our Factory
Our Factory

Quality Testing of the Product
Our product undergoes strict testing for tensile strength, corrosion resistance, and biocompatibility to ensure reliable performance in joint implants. We use advanced methods to verify its safety and durability, and all products meet international medical standards for consistent, long-term use in the body.


FAQs on Cobalt Chromium Tube
1.What is the main advantage of cobalt chromium alloy over stainless steel in stent platforms?
CoCr alloy offers higher strength, allowing thinner struts for better flexibility, deliverability, and reduced restenosis risk while maintaining excellent radial strength and radiopacity in drug-eluting stents.
2.Is this tube biocompatible for long-term implantation?
Yes — medical grade cobalt chromium demonstrates outstanding biocompatibility, low thrombosis rates, and minimal ion leaching, as validated in extensive FDA-reviewed studies on implantable medical devices and cardiovascular stents.
3.Does the CoCr tube support thin-strut drug-eluting stent designs?
Absolutely. Its superior mechanical properties enable ultra-thin struts (e.g., 60-80μm range), improving endothelial coverage and lowering neointimal formation in modern drug-eluting stent platforms.
4.What standards does this medical grade tube comply with?
Conforms to ASTM F562 (or equivalent L605) specifications for wrought cobalt-chromium-nickel-tungsten alloy, ensuring quality for implantable medical applications.
5.Can this tube be used for custom or peripheral stent manufacturing?
Yes — its precision seamless form and customizable dimensions make it ideal for both coronary drug-eluting stents and peripheral vascular applications requiring high fatigue resistance and corrosion protection.










