Overview of Platinum Iridium Medical Tube
Our ASTM B684 Platinum Iridium medical tubes deliver exceptional performance in neurostimulation and implantable medical devices. Manufactured from high-purity platinum-iridium alloys (commonly 90Pt/10Ir or 80Pt/20Ir), these precision medical tubes combine outstanding electrical conductivity, mechanical durability, and biocompatibility. They are ideal for long-term implantation in neuromodulation systems, offering low impedance, high charge transfer capacity, and excellent resistance to bodily fluids. Whether used in deep brain stimulation (DBS) leads, spinal cord stimulation (SCS) electrodes, or custom hypotube components, these biocompatible tubes ensure reliable signal transmission and structural integrity in demanding biomedical environments.


Features of ASTM B684 Platinum Iridium Alloy Medical Tubing
Exceptional electrical conductivity and low impedance for efficient neurostimulation signal delivery in implantable electrodes and leads.
Superior corrosion resistance and long-term biocompatibility, proven safe for chronic implantation in neural tissue.
High radiopacity for precise visualization under fluoroscopy and medical imaging during neurostimulation procedures.
Precision manufacturing with tight tolerances, smooth finishes, and customizable diameters/wall thicknesses for complex hypotube assemblies.
Full compliance with ASTM B684 standards for platinum-iridium materials, ensuring consistent quality in medical-grade tubing.
Balanced mechanical strength, hardness, and flexibility from iridium alloying, supporting minimally invasive delivery in neuromodulation systems.
MRI-conditional compatibility in most neurostimulation configurations, enhancing patient safety and post-implant imaging options.


Applications of Platinum Iridium Medical Tubes
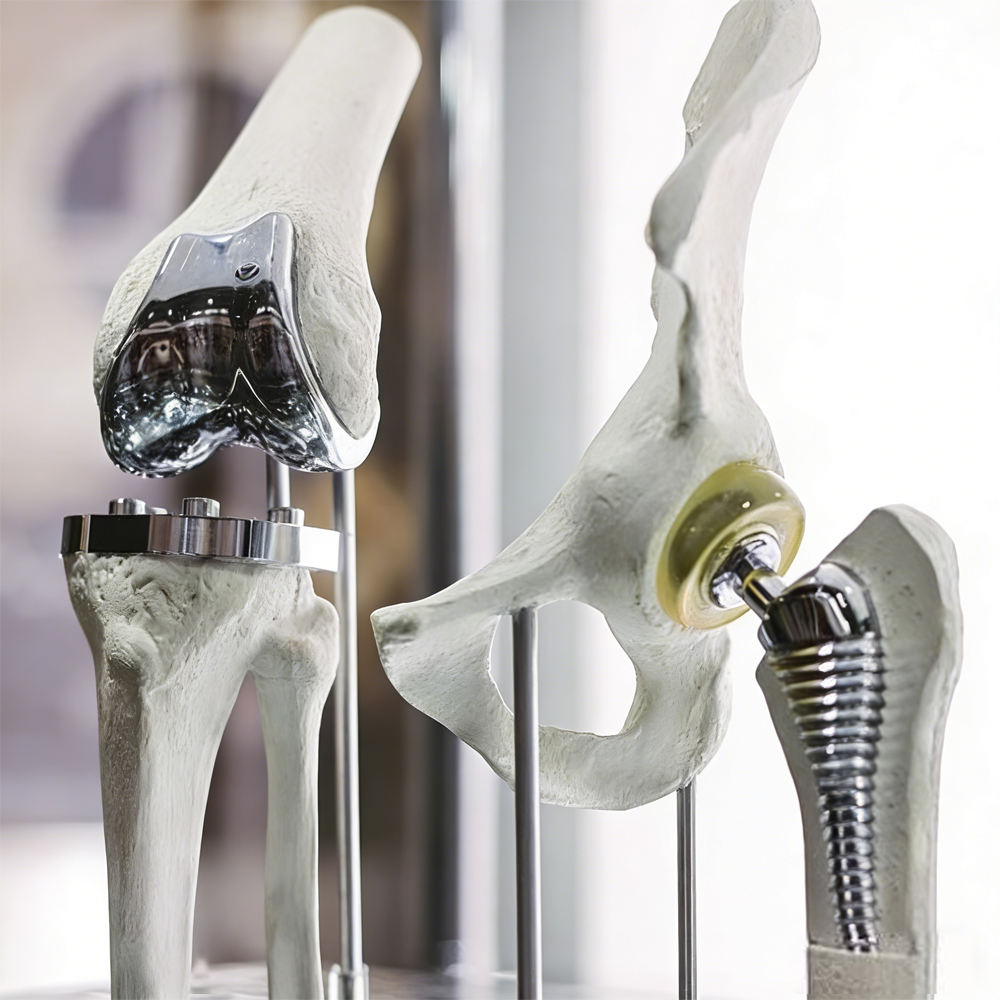
Cardiac Rhythm Management Devices — Pacemaker leads, ICD leads, and feedthrough components requiring reliable electrical performance and long-term stability.
Neuromodulation and Neurostimulation Systems — Deep brain stimulation (DBS) electrodes, spinal cord stimulators, and other implantable neurostimulator leads.
Cochlear and Auditory Implants — Electrode arrays and wiring in cochlear implants demanding high fatigue resistance and biocompatibility.
Radiopaque Marker Bands and Stent Components — Precision-cut tubing for vascular stents, catheters, and minimally invasive interventional devices.
Sensor Housings and Diagnostic Implants — Components for continuous glucose monitors, pressure sensors, and other implantable diagnostic systems.
Custom Lead Wires and Electrophysiology Components — Tailored solutions for research-grade and commercial implantable medical electronics.
ABOUT US
Victory Alloy Co., Ltd. is a professional manufacturer specializing in medical alloy materials, with a dedicated focus on the research, development and production of high-performance biomedical alloys.
Our primary product range includes medical-grade titanium alloys, Nitinol (nickel-titanium alloy), cobalt-chromium (CoCr) alloys, platinum-iridium (Pt-Ir) alloys, and medical stainless steel. These high-quality alloys are widely applied in a broad spectrum of medical fields, such as cardiovascular stents, orthopedic implants, interventional guidewires, dental implants, surgical instruments, and disposable medical auxiliary devices. Boasting outstanding biocompatibility, strong corrosion resistance and excellent mechanical properties, our products have won consistent praise from customers across the board.

Client Visits
We welcome client visits to showcase our manufacturing and testing processes. Clients can explore our facility, discuss specific needs, and learn about our quality control methods. Our team ensures a personalized experience to meet your exact requirements for medical applications.

 Our Factory
Our Factory

Quality Testing of the Product
Our product undergoes strict testing for tensile strength, corrosion resistance, and biocompatibility to ensure reliable performance in joint implants. We use advanced methods to verify its safety and durability, and all products meet international medical standards for consistent, long-term use in the body.


FAQs About Medical Grade Platinum Iridium (Pt90/Ir10) Tubes
Q: What does ASTM B684 compliance mean for medical tubing?
A: ASTM B684 specifies high-purity platinum-iridium alloys (such as 90/10 or 80/20 Pt/Ir) for electrical contact materials. When applied to medical tubes, it guarantees consistent composition, excellent conductivity, and reliability for neurostimulation leads and implantable devices.
Q: Why is platinum iridium the preferred alloy for neurostimulation electrodes and tubing?
A: Platinum iridium offers unmatched biocompatibility, corrosion resistance in bodily fluids, low impedance, high charge injection capacity, and added mechanical strength from iridium—making it superior for long-term neural stimulation compared to stainless steel or other alloys.
Q: Are these platinum iridium medical tubes suitable for chronic implantation?
A: Yes. They are widely used and FDA-cleared in many neurostimulation systems (DBS, SCS, etc.) due to their proven long-term stability, minimal tissue reaction, and resistance to degradation.
Q: What customization options are available for the tubing?
A: We provide various outer/inner diameters, wall thicknesses, lengths, alloy ratios, surface finishes, and secondary operations (e.g., laser cutting, electropolishing) to match your specific neurostimulation lead or implantable device requirements.
Q: Is the platinum iridium tubing MRI compatible?
A: Most configurations are MRI conditional. Always verify compatibility with the final assembled neurostimulation device labeling and consult regulatory guidelines for your specific implantable application.











